Precision Process. Unified Workflow. Compliant Results.
Where medical expertise meets a validated, secure linguistic environment.
How We Work: Precision Translation Process
Our validated How We Work process sets the quality standard for every service we offer, from clinical protocols to informed consents and other patient-facing documents.
All translations are performed by expert human linguists within a secure platform that integrates productivity and precision tools, following a rigorous 8-step QA process to ensure clinical accuracy, consistency, and regulatory compliance.
BioLingo Connect is the secure, HIPAA-compliant platform that supports this workflow, serving as a central hub for project management, workflow visibility, and document version control, with full transparency, traceability, and audit-ready documentation.
Ready for compliant medical translation?
Get Your Free Quote
Project scoped and priced in 24 hours.
8-Step QA PROCESS
BEFORE TRANSLATION
Define Your Needs
We review your document type, target audience, regulatory context, and required literacy level to define project specifications before translation begins.
Expert Team Selection
We select the translation and editing team that best suits the subject matter and expertise needed for your project.
Glossary and Style Guide
We establish or integrate client-specific glossaries and terminology controls to ensure consistency across studies, sites, and ongoing projects.
TRANSLATION & REVIEW
Translation Platform
All translations are completed within our secure, encrypted translation environment with terminology validation, version control, and integrated QA checkpoints. This ensures consistency, traceability, and controlled document handling.
Editing
A second medical linguist independently reviews the translation for clinical accuracy, terminology consistency, readability level, and adherence to project specifications.
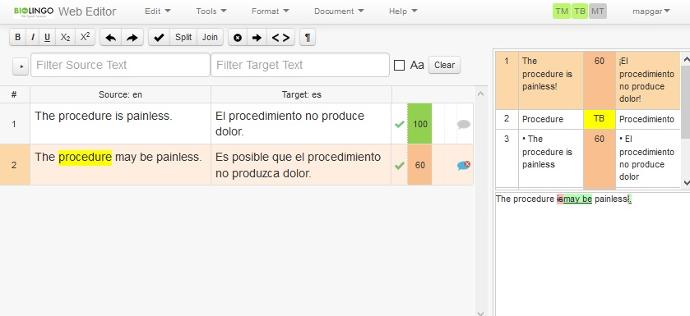
Proofreading
A final proofreader conducts a quality control review for formatting integrity, linguistic precision, and typographical accuracy prior to release.
Desktop Publishing
We ensure layout accuracy, text expansion management, and formatting consistency to preserve the integrity of patient-facing and regulatory documents.

Delivery and Follow Up
Final documents are delivered securely along with documentation required for institutional or IRB review. Ongoing terminology updates are incorporated into future projects to maintain long-term consistency.
Digital Oversight & Workflow Transparency
Every stage is managed within BioLingo Connect, bridging linguists and stakeholders
with full visibility into your project lifecycle.
Centralized Ordering
Securely upload sensitive clinical data.
Real-Time Tracking
Monitor your project’s progress through every stage of the QA process.
Audit-Ready History
Access every version and invoice in one organized location.
The Engine of Consistency: Integrated Linguistic Assets
Specialized tools embedded in our translation platform preserve your knowledge and optimize costs.
Our full service capabilities
40+ languages, 50+ formats, certification options
Life Science Translation FAQs
Is this 8-step process followed for every project?
Yes. Whether it is a 1-page medical report or a 500-page regulatory filing, our "8-Step Model" is our standard operating procedure to ensure defensible quality.
Do you provide proof of this process for our audits?
Yes. We provide Certificates of Accuracy for every project and maintain a full audit trail within BioLingo Connect, documenting every linguistic step and quality checkpoint.
How do you handle "Layperson" language for patients?
During Step 1, we define the "literacy target." Our editors in Step 5 specifically check to ensure that complex medical jargon has been adapted into clear, understandable language for the intended audience.
How does BioLingo safeguard Protected Health Information (PHI)?
All data handling occurs within BioLingo Connect, which follows strict HIPAA-compliant protocols. We use high-level encryption for files at rest and in transit, and every translator or subject-matter expert operates under a comprehensive NDA to ensure total confidentiality.
Does BioLingo use Machine Translation (MT)?
For clinical, regulatory, and patient-safety content , we do not utilize raw machine translation. These high-stakes documents are translated and reviewed exclusively by human medical subject-matter experts.
However, for non-technical or internal-use content , we do offer an MTPE (Machine Translation Post-Editing) workflow. This combines the speed of MT with a mandatory layer of human review to ensure the final output is accurate and coherent while remaining cost-effective for lower-risk materials.
Accurate. Compliant. IRB-Ready.
Launch your next translation project today.
Get My Quote